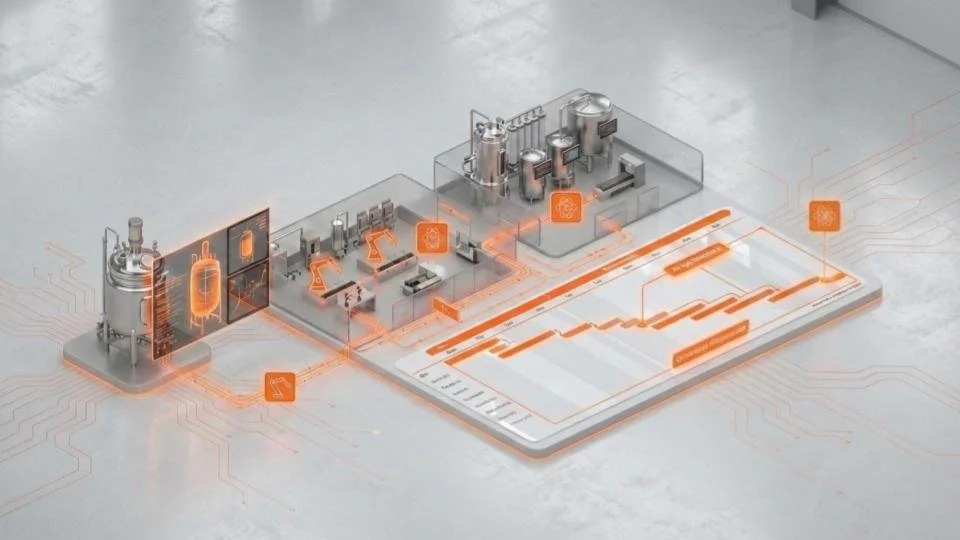
Accelerate development. Strengthen compliance. Scale with confidence.
Build a next-generation technology backbone for CMC and Process R&D; one that embeds AI, automation, digital twins, and intelligent process control across development, scale-up, and manufacturing.
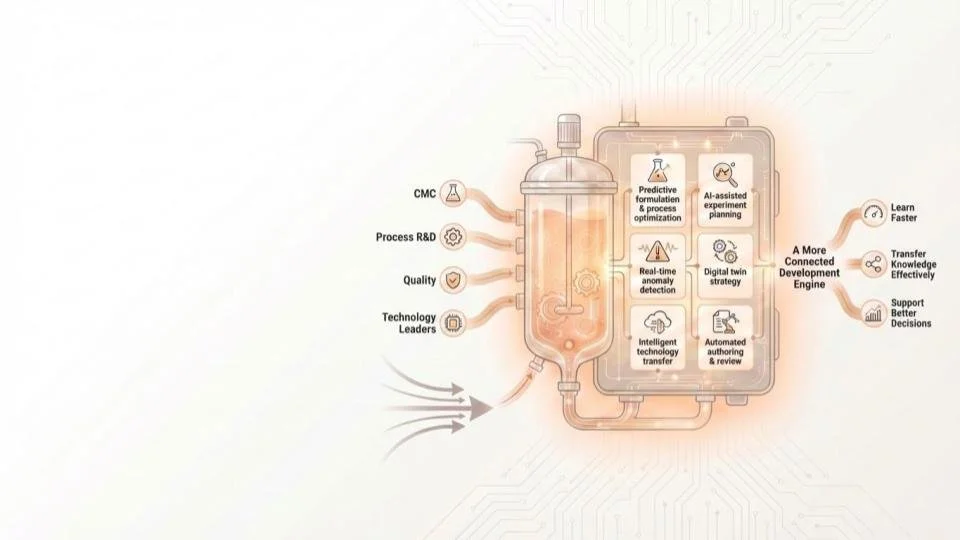
What we help biopharma & CDMOs achieve
We partner with CMC, Process R&D, Quality, and Technology Leaders to identify high-value AI opportunities and make them production-ready
The result is a more connected development engine that can learn faster, transfer knowledge more effectively, and support better decisions across the product life cycle.
Why this matters now?
CMC and manufacturing teams are under pressure to shorten development timelines, improve robustness, and reduce manual work while maintaining strict compliance
That creates a major opportunity to unify data, models and workflows into a more self-optimizing environment
Our focus areas include:
AI strategy for CMC and Process R&D.
Manufacturing data architecture and knowledge graph design.
Digital twin and process simulation advisory.
GenAI copilots for scientists, engineers, and technical writers.
Agentic workflow design for lab, development, and tech transfer tasks.
Model governance, validation, and GxP-ready implementation support.


Assess
We evaluate current CMC workflows, systems, and pain points to identify the fastest paths to value.
Design
We define the use-case portfolio, data architecture, governance model, and operating rhythms required for success.
Pilot
We stand up targeted pilots that demonstrate clear impact in areas such as process optimization, document generation, or digital twin-enabled decision support.
Scale
We help expand successful pilots across programs, sites, and functions with the right controls for validation, auditability, and adoption.
Let’s build the next-generation of CMC
AI in CMC must be useful, trustworthy, and auditable. We design solutions that respect data integrity, validation, human oversight, and regulatory expectations from the start. That means practical adoption, not experimental novelty.
Benefits of integrated process development & scale-up
Faster process development and scale-up
Better reuse of historical knowledge
Reduced manual documentation burden
Earlier detection of process issues
Stronger tech transfer outcomes
A clear path from pilot to enterprise scale